Friday, July 20, 2007
Automatic Segmentation: Some Ideas
1. Users like control
Currently, the algorithm developed by Noel Codella is highly automated, but in doing so, takes much of the control away from the user. To operate the program, the user must simply identify the left ventricle in one slice of the cine MRI with a single click. The algorithm handles the rest. But to an experienced clinician who is used to performing all of the tasks manually, this degree of automation is a little disconcerting. I believe adding an option for the user to "tweak" the algorithm's segmentation decisions would lessen this sense of detatchment. While it is debatable whether a computer or a human can provide the best segmentation decisions, giving the option to perform manual corrections would make the user far more comfortable in using the program.
2. Users like feedback
As much as everyone loves to hate the little status bars the creep across the screen while we perform tasks on the computer, they are essential. I never really realized this until I saw the automatic segmenter in action. The program, while relatively fast, requires up to 30 to 45 seconds to fully complete segmentation after the initial user input. Unfortunately, there is no visual indication concerning the program's progress during this time. In every trial thus far, the program has finished successfully every single time it has been run, so it would seem that a status bar would serve little purpose other than to provide a little visual entertainment for the user. In reality, I've found that this has been one of the major complaints about the program's current implementation. Users like feedback about what the computer is doing!
These are all pretty minor suggestions that have little to nothing to do with the algorithm itself, but I feel changes such as these would make the program far more user friendly and will make it more likely for the program to be used in an actual clinical setting in the future.
Thursday, July 19, 2007
Hearts and Lungs


Wednesday, July 18, 2007
Cranioplasty with Insertion of Medpor
So, now for some details…first, an incision was made from ear to ear across the crown of his head. The face was eventually peeled forward to reveal the defect and then the neurosurgeons, wearing super-geeky, thick double-lensed, black-rimmed glasses, started picking his brain…the not-so-fun, invasive way. I believe all they were doing was taking biopsies as very little tissue was removed, but I could very well be mistaken. It was so cool. There I was gawking at this guy’s brain pulsing to the rhythm of his blood flow. Awesome. It was actually quite curious, the location of the defect and extent of the face peel really looked like his eyeballs should have been starring back at me…yet, they weren’t…weird. I guess they were hidden under his face flapped over his nose. That sounds icky.
Anyway, tag team high five and plastic surgery’s on the scene. The procedure began by a leisurely drilling of his skull to isolate a portion of cranium destined for an autograft. Bone filings went EVERYWHERE…the surgeons’ faces, the floor, half of the observers - everywhere. Following this mess was a rather barbaric technology (if you ask me)…the hammer and chisel. Yes, we can put a man on the moon, genetically engineer all sorts of things, and yet, in order to take a simple graft we still use the age old, chisel to beat on someone’s skull until chips of bone fly into a nice warm, blue bowl of saline…there’s gotta be a better way to harvest bone grafts. This dude took a beating…again. So, just for a visual (since the pictures I took were on the fellow’s camera, and thus, gone forever), this guy’s face is peeled off, his parietal skull bone is drilled and chiseled away to remove a bone graft and blood is everywhere...and he's still missing most of the frontal bone.
Now for the cool innovations. First, a material developed by the medical device company, Stryker, provided us with a quick-drying (~30-40 seconds) cement of hydroxyapatite…a precursor to bone. This paste was spread over the donor site created on the parietal bone…and eventually along the interface of the implant and the skull. The next cool biomaterial used was Medpor (the implant), which is a custom made, perfectly contoured, porous sheet of high-density polyethylene microspheres sintered together...kinda like the small piece shown below.
Medpor is used instead of say, PMMA (polymethylmethacrylate), due to the lower risk of complications. Potential side effects of PMMA may include local tissue damage caused by its exothermic reaction, release of toxins, and the possibility for a high rate of infection. Medpor doesn’t have these. However, Medpor has several very enticing benefits, including its flexibility and ease of coverage, its biocompatibility, and its porous nature, allowing for the rapid ingrowth of vasculature and soft tissue. The latter is very important because it promotes wound healing and stabilizes the implant.
As an aside, one study I read about had zero post operative infections, zero wound breakdowns, and zero follow up surgeries after a period of 4 years in 611 procedures (Liu et al., Neurosurg. Focus 16, March 2004). Pretty cool stuff. I hope I invent something totally sweet like this.
Anyway, this sheet of Medpor flawlessly fit into the man’s cranial defect and was screwed into the skull for support with titanium (inert and biocompatible) screws. It looked something like this, but this is not from the actual case.
To finish the case, the man’s face was stapled and sewn back on…and despite the head bandage, he looked pretty normal. Amazing.
tying a knot laparoscopicly
The patient had a retroflex sigmoid colon. Because of gravity, the redundant colon constantly drags other part of colon and pushes rectum, which also brings pain to the patient. What the doctor will do is that he will use threads to knit a net to pull up the redundant colon. It sounds like a good idea.
At the begi
 nning, the doctor made several small incisions on the abdomen and put come metal pipes through the abdominal wall as channels, so that a laparoscope and some other surgical devices could easily go in. Then CO2 was inflated and abdomen was blown up like a balloon to provide working and viewing place. The laparoscope captured real time movies and transmitted it to a high definition LCD. This is doctors’ “eyes” in the operation. Those long thin surgical devices are doctors’ “hands”.
nning, the doctor made several small incisions on the abdomen and put come metal pipes through the abdominal wall as channels, so that a laparoscope and some other surgical devices could easily go in. Then CO2 was inflated and abdomen was blown up like a balloon to provide working and viewing place. The laparoscope captured real time movies and transmitted it to a high definition LCD. This is doctors’ “eyes” in the operation. Those long thin surgical devices are doctors’ “hands”.Then we began a wild exploration. After identified several organs, the doctor thought that the position of uterus was hindering the operation so he delicately tacked the uterus onto the abdominal wall. He afterwards reassured me that considering the patient’s age, the chance for her to use her uterus was extremely low. That’s the reason he did so and it wasn’t a common procedure.

Finally, the retroflex colon was found. What happened next was I saw needles and threads going into the metal pipe. The long thin picker was so agile and efficient in such a small space. Within less than half an hour, some knots magically shown up on the monitor, and gradually assembled into a net. The redundant colon was fixed.
When the doctor was tying a knot, he tried to convince me that making a knot laparoscopicly is simple. I don’t believe him this time. Why? Just check out the underneath animation.

Neurological Surgery
This past week, I had the chance to observe several more procedures in the OR, including observing a kidney transplant surgery with Dickson. I watched the removal of the kidney from one patient, handling of the kidney while the recipient was opened up, and attachment of the kidney into the recipient. The connections that must be attached when put in the recipient are shown in the figure. I found the handling of the kidney external to the human body to be especially interesting. Since there were delays in opening up the recipient, the transplant doctor kept the kidney in slush. Then, when the recipient was ready to receive the kidney, the doctor wrapped the kidney in some wet cloths packed with slush in what he described as a ‘sushi roll’. He used this as an ice pack to keep the kidney cold while attaching the kidney to the patient and mentioned the need for a better kidney preservation machine that could mimic the human body and would not require cooling the kidney down.
Neurological Surgery Aneurysm Treatment
As I’ve mentioned previously, there are two major techniques to treat brain aneurysms – clipping the aneurysm or using filling the aneurysm with coils through endovascular means, also known as coil embolization. Coil embolization is much more attractive because it is less invasive, however there are limitations on when coil embolization can be used, depending on aneurysm shape, as well as problems with aneurysm recurrence (defined as aneurysm neck enlargement or recanalization) when the aneurysm is not adequately occluded.
Figure illustrates filling of aneurysm with coil. www.brainaneurysm.com
Traditionally, coils have been made out of platinum. One popular coil is the gugliemi detachable coil (GDC) which has a shape memory effect. It is routed into the aneurysm and detached from a pusher wire using a low current. Due to the problems demonstrated with significant recurrence rates, researchers have been trying to come up with techniques to increase the percent occlusion (defined as the volume of the coil over the volume of the aneurysm). A modification of the GDC coil was introduced, called the Matrix coil, which has an absorbable polyglycolic-polylactic acid (PLGA) copolymer coating on platinum coils. The PLGA is absorbed by the body within 90 days. The premise for why this technology is an improvement over GDC is that it provides a much higher initial occlusion. I am working on analyzing clinical data to investigate and compare the outcomes of the different treatments.
Figure illustrates mechanism for inserting matrix coils as well as different configurations of coils. www.bostonscientific.co.uk
Portable X-Rays Acquisition

Above are some portable scanners to give you an idea of what they are like. For reference the ones I saw were specifically GE Medical Solutions. And Kodak makes the film (Surprise! or not). From a technical point of view, portable X-ray machines are actually rather intriguing. They don't expose film that a person can read right away. Instead you shoot at a cartridge (a $400 re-usable cartridge) that you place behind a person while the are laying or sitting down. The cartridge is basically comprised of a box with a phosphor plate inside. Once the scan is acquired, you pass into a machine and it gets scanned into the hospitals PACS (picture archive effectively). Then it can be passed to radiology for reading digitally, no film running here! Also, a portable X-ray machine has a far more concentrated beam and much less scattering, so no lead coats are required, which is useful.
So I traveled the 4th floor with the tech (nice guy), and got to see how he acquired the scans. Most people are very understanding when he comes in (I think its because of all medical procedures they understand x-ray the most). The tech usually leans the patient forward, slides the plate under their chest, steps 70" back, yells "X-ray", hits a button, yells "Clear", and we are done. Its quick. The general result is below

Sometimes there are problems. Like if a doctor doesn't put in orders, but need it done 'stat'. Or if there is double-exposure. Or the patients obese. Or the patient is rotated. For that last one sometimes there nothing that can be done.
da Vinci...man was a genius


The da Vinci surgical system consists of two major components. The first component is comprised of three robotic arms (top image). The center arm is used to control the endoscope and the other two arms control the two instruments. The other major component is the control center (bottom image). For this procedure, 5 incision points are made in the abdomen of the patient. One incision point in made near the medial line of the body, which is where the endoscopic arm will be inserted. Two more incisions, one on each side of the middle incision, will be made for the two robotic arms that will control the surgical instruments. And two final incision points will be made on the abdominal sides, for two additional instruments used by the nurses to assist in the surgery. Below is an image of what whole procedure may look like.

Throughout the surgert, the doctor will sit at the control station and control the arms. It was truly an awesome sight to see the large robotic arms swaying around and hitting the assisting nurses as the instruments cut through the tissue. Below is a picture of what one of the tools may look like. The precision that the system allows is incredible. One of the main issues of the surgery is to not cut the nerve that may be responsible for sensation. This system allows the surgeon to clearly see what he/she is cutting.

In the end, the prostate is disconnected from the bladder, the seminal vesicles and vas deferens are clipped, and the urethra is reattached to the bladder. The man will obvioulsy not be able to have children, but if the surgery went well, he would still be able to have sensation in his penis and will still be able to have an orgasm, although there would be no ejaculation. Leonardo didn't design this system, but if he were still alive today, I know he would have.
Tuesday, July 17, 2007
Esophageal Atresia Surgery
But first I’ll explain the background once again. EA and TE fistula is a congenital disease where the esophagus and trachea do not separate. Instead, you can get variations of this disease. For example, most patients (86%) have the variation when the upper esophagus pouch ends close to the trachea and a fistula is present at the distal end of the trachea connecting to the esophagus.
http://www.aafp.org
Other variations include no fistula, or only a fistula. You can get a proximal TE fistula or even a double fistula at the proximal and distal ends. These variations are rare and make up for the last 14% combined.
Now for the surgery:
The procedure is relatively simple, as the mortality rate is much lower than in the past.
http://www.answers.com/topic/esophageal-atresia-repair?cat=health
To repair esophageal atresia, an opening is cut into the chest (A). The two parts of the existing esophagus are identified (B). The lower esophagus is detached from the trachea (C) and connected to the upper part of the esophagus (D). The wound in the trachea is closed, and the chest incision is repaired.
In our case, an interposition colon procedure was unnecessary. The patient's esophagus was able to overcome the wide gap and connect together. This is called an esophagoesophageal anastomoses. Otherwise, a section of the colon will have to be placed in the gap between the two ends of the esophagus. The procedure went well and the patient is recovering.
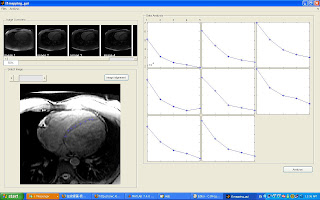
T1 Mapping GUI
http://www.e-mri.org/cours/Module_4_Signal/ponderations4_en.html
As mentioned in the first week, in diagnostic ischemic heart disease, one important factor is to distinguish infarcted myocardium from viable myocardium. The ideal imaging method will clearly distinguish these two types of myocardium as shown below.
Using DE-MRI, this is achievable because Gadolinium injection makes infarcted myocardium having a much shorter T1. This results in infarted myocardium shows up bright while normal myocardium shows up dark. However, we need to acquire data at the right time to achieve the desired contrast. As shown in the figures below, if we acquire data at inappropriate time, we can not get the contrast as we want.
Therefore an imaging analysis tool is developed for T1 mapping analysis, the gui is shown below:
DiVinchi Pictures from the cell phone

Patient with the DiVinchi arms penetrating his abdomen. The doctor at this point was cutting the patients urethra and epedidemis using cutlery and crimps.
Dr. Tewari is hard at work using the DaVinchi interface with the robotic arms. He does not even need to scrub in!
Oh I forgot to mention...this system is around $1 million, and I believe the Dr. is trying to get an upgrade system so he can perform even more of these procedures.
Nuclear Imaging in Cancer
The positron emission tomography (PET) scan creates computerized images of chemical changes, such as sugar metabolism, that take place in tissue. Typically, the patient is given an injection of a substance that consists of a combination of a sugar and a small amount of radioactively labeled sugar. The radioactive sugar can help in locating a tumor, because cancer cells take up or absorb sugar more avidly than other tissues in the body.
After receiving the radioactive sugar, the patient lies still for about 60 minutes while the radioactively labeled sugar circulates throughout the body. If a tumor is present, the radioactive sugar will accumulate in the tumor. The patient then lies on a table, which gradually moves through the PET scanner 6 to 7 times during a 45-60-minute period. The PET scanner is used to detect the distribution of the sugar in the tumor and in the body. By the combined matching of a CT scan with PET images, there is an improved capacity to discriminate normal from abnormal tissues. A computer translates this information into the images that are interpreted by a radiologist.
PET scans may play a role in determining whether a mass is cancerous (under clinical trial). However, PET scans are more accurate in detecting larger and more aggressive tumors than they are in locating tumors that are smaller than 8 mm and/or less aggressive. They may also detect cancer when other imaging techniques show normal results. PET scans may be helpful in evaluating and staging recurrent disease (cancer that has come back). PET scans are beginning to be used to check if a treatment is working - if a tumor cells are dying and thus using less sugar. I put together an example of a case where CT fails to detect cancerous nodules while PET scan detects them.


HISTORY:
65 year old with recent right lower lobe resection referred for follow up.
IMAGING FINDINGS:
Increased metabolic activity consistent with recurrent malignancy in the right hilar region, L4 vertebra and liver. None of these lesions are well visualized on the CT portion of the exam, however, these lesions are compatible with metastatic disease.
DISCUSSION:
This is an unfortunate case of a man who underwent right lower lobe resection for a primary squamous cell carcinoma (non small cell). Six months later he was referred for PET•CT which showed at least 4 metastatic lesions. Unfortunately, this patient did not have a preoperation PET•CT which likely would have shown at least one of the metastatic lesions and made the patient a non-operative candidate. Interestingly, all four metastatic lesions were not able to be seen on the CT portion of the scan. The liver lesion wasn't well visualized on the CT portion of the exam either even with a good IV contrast enhanced exam.
My week in Cardiothoracic Surgery
Coronary Artery Bypass Graft (CABG) surgery
Approximately 500, 000 cases are performed in the U.S every year. The primary of cause of Coronary artery disease (CAD) is the deposition of cholesterol around the coronary artery, causing atherosclerotic plaque (hardening of the arteries). Other causes including high blood pressure, elevelated cholesterol, and diabetes.Cardiosurgeon at Work...

Once the patient has been put under anesthesia and intipated, the anesthesiologist inserts leads for echocardiogram to monitor the heart function and another set of lead electrodes to monitor brain function. Monitoring the brain and renal function are essential ensuring that the patient do not suffer significant ischemia once the heart(myocardium) has been stopped..After the pericardium(heart sac) has been exposed and the location to be operated identified, blood from the vena cava( de-oxygenated) and the blood from the lungs(0xygenated) are re-directed to the bypass machine. The heart is then injected with high K solution to stop it. The bypass blood is maintained at a certain perfusion pressure and the blood cooled down to 25 degree to reduce metabolic activity and reduce ischemia to the brain. Again, renal function is quite indicative of any significant ischemia. So before the surgeon operates on the heart, it is completely stopped and chilled to 4 degree with ice slush. You can see the myocardium contract slightly after it has been cut...Really cool!
In the figure below, the blood is cannulated by tapping into the topmost superior vena cava and the aorta.

At a 40-min time interval while the heart is stopped, it is injected to high K solution to 'sedate' it. The heart ideally should not be injected over 3 times which means that the surgeon has to try to stay within a 120-min window within which the heart has been stopped. Everything is hectic during this time, with the surgeon instructing all the concerned to carry out different tasks, warm the body, add anti-coagulants, increase perfusion pressure, and etc...
Once the grafts have been sutured, the heart and the body are warmed to the physiological temp. and then heart is resuscitated by shocking it. At this point, it is a matter of ensuring that the heart beats properly before the blood is redirected from the bypass machine and the patient is sutured to ensure there is no bleeding. I couldn't believe seeing the heart beat again..wowoow. I will blog about the valve replacement in my next blog..
Dickson
Monday, July 16, 2007
Robot Assisted Surgery
I will have some pic's up later but I was able to watch in 3D the removal of a prostate in a 60-70 year old male patient. The setup was pretty simple: ports were placed into the pelvic area, then the robot was placed over the patient, and the rest the Dr. did about 7 ft away from the operating table...manipulating the robotic arms via the robot/human controls!
The nature of the surgery is truly robot assisted though, because multiple Drs are still required to aid the robotic arms with other laproscopic tools near the bed. However the robot gives the dr doing the majority of the "cutting/surgery" many more degrees of freedom in manipulating the laproscopic devices, thus he can cut, trim, sutcher, pull, etc. etc. in many different , human impossible angles.
Very very cool biomedical engineering.
One Patent-Pending: Chatting with histology department
These Dr.'s see multiple tissue samples a day, hunching over the microscope, severely straining back-neck-and-shoulders by the end of the day. One of the greatest improvements a resident wished for was a automatic remote controlled slide mover, and fiber-optic goggle attachment so that he may look at the specimen while sitting back in his chair. (I did a quick USPTO search....then submitted a provisional patent application) This would let histology look at many, many samples while relaxed!!
Also: I found out that you may test positive for TB if you look at to many TB-slices (e.g. most of the residence/Drs now test + at Weill) kinda scary!
One more histology fact: Fixing tissue is much easier to look at then, fresh or frozen tissue!
As with anything, there are no guarantees...

Another patient we saw, had a tumor that grew fives time its size from 5 years ago. The figure above shows an MRI scan of a brain tumor in the brain. Here the tumor looks white. This is becuase the tumoregenic tissue produces high contrast relative to normal braint tissue. This patient had done extensive research and knew what he was getting into. What was different about this case was that his family was in the room with him. It was a very intense situation for me, to see how the family was reacting to the prognosis of his condition. No matter what numbers my clinician was giving him, he didn't seem to understand that nothing is guaranteed. I believe he was going to get back to my clinician on what he planned on doing. One of the more interesting aspects of this case was how adamant the patient was on getting the statistics of his outcome. To me, the prognosis was obvious: you have a huge tumor in your head, take it out or else you will definitely lose cognitive function. Pecentages are all relative.
Neuro Detour
Week 4
This week I took a detour from vascular surgery to watch a couple of neurological surgery cases. One really interesting case was the removal of a pituitary tumor.
Removal of Pituitary Tumor
Endoscopic Endonasal Surgery
http://www.skullbaseinstitute.com/video_pituitary_tumor.htm
There is a great video of the procedure at the link above. A few stills are shown below:
 The endoscope is inserted through the nostril and navigated to the brain.
The endoscope is inserted through the nostril and navigated to the brain.
The dura (covering of the brain) is incised.

The tumor is gradually removed.
Source: http://www.skullbaseinstitute.com/video_pituitary_tumor.htm
Research Project
I’ve also been working on my research project regarding the outcomes of vascular procedures in women. Some of the demographics of the telephone interviews I have been conducting are shown below:

This chart shows the proportions of women I have contacted that are on or have had hormone replacement therapy (HRT), have been diagnosed with osteoporosis, take osteoporosis supplements, and who take calcium or vitamin supplements (total does not equal 100%). I’m still working on the data analysis regarding the outcomes of procedures and hope to have results soon!
Sunday, July 15, 2007
From Research to the Real World
As I mentioned in my previous post, my research topic is on esophageal atresia (EA) with or without tracheoesophageal fistula (TEF). The disease is a congenital underdevelopment of the esophagus and trachea. These two tubes, for the most part, can be connected to each other, making eating and breathing almost impossible for the patient. The disease is not rare among newborns and has a prevalence of about 1 in 4000. For the past few weeks, I have just been reading up and doing statistical analysis on past cases on EA/TEF. To my surprise there happened to be a patient born and admitted in the neonatal intensive care unit (NICU) a few weeks ago with EA/TEF. I took the time to check out the patient and the patient history as my research and the real world were finally integrated.
umd.edu
The patient was born with the most common type EA/TEF. Her upper esophagus discontinued into a pouch and her bottom esophagus connected to her trachea (which would be the fistula part). As of recent decades, this repair would be an easy fix with a low mortality rate. Normally, the patient would be operated on within the first 48 hours, which is known as primary repair. However, this patient, and like 30% of all EA/TEF patients, has some type of other complication. In this case it was cardiac problems. The patient had to be admitted into surgery to fix her heart problem before fixing the EA/TEF. She would not be able to survive the EA/TEF recovery without everything else being healthy.
I wasn’t there for her heart operation, but found out that when the doctors closed the patients chest back up, the heart was beating against the chest wall causing too much pressure. The doctors, then had to re-open the chest and leave it open for a few days until she stabilized from heart sugery. They used some type of plastic see-thru-wrap (much like cling-wrap) to close up the chest cavity from the outside environment. This allowed the heart to beat freely. She remained in the NICU until her body stabilized; upon stabilization she returned to the operating room to have her chest closed. She will be back to the operating room in a few days (weeks?) to have her esophagus disconnected from her trachea and to connect the two disconnected esophagus’. They are waiting for her to stabilize before performing this operation.
In retrospect, the complications the patient had with her EA/TEF would have led to a low survival rate only 10-20 some years ago. Over the past decade the overall mortality for EA/TEF have gone down to 11% from 22%. This is due to refinements in NICU care, anesthetic management, ventilator support, and improved surgical techniques. The corrective surgery for EA/TEF is at a 96-98% success rate presently, but was at a 0% survival rate just 50 years ago.
As researchers especially in academic research, we do a lot of basic science –discovering this, studying about that. A lot of our research won’t make a difference in the real world until decades from now. This other realm of research I’ve been exposed to has some gratifying feelings, that being once you’re done with your research, it will have a direct impact on people and their lives.
4th Week in Plastics
I started off the past week with a Monday morning lecture on lower extremity wounds. Dr. Spector gave the lecture which was followed by case reviews for the residents. This was very interesting to watch because the reviews allowed insight into the entire diagnosis and treatment process; describing from when a patient presents with a particular case, noting what knowledge is important to gain about the patient in order to make a proper decision, and finally how to treat the patient and what should be done when obstacles are confronted.
During office hours I saw a burn patient that Dr. Spector has been treating. The patient has scaring all over his face and upper torso which is disfiguring, painful, and prevents proper function. Around his mouth and neck the scarring has hardened so that he is not able to fully open is mouth or rotate his head. In addition, his nose is no longer symmetric which Dr. Spector plans to alter by removing cartilage and skin from his nose to reform the nostril. In addition, I saw quite a few cosmetic consults during office hours where Dr. Spector discusses the desired alterations a patient may want to his or her body including breast augmentations, rhinoplastys, or liposuction. I also met a patient that Dr. Spector performed a breast augmentation for a few years ago and recently one breast has enlarged and become painful as the fibrous tissue scarring has begun to contract. This is a known complication to breast augmentation. In order to fix the problem, the patient will have to undergo surgery to remove the fibrous tissue.
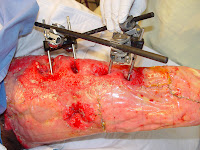
I have also seen some interesting cases in the OR. Prior to my arrival, a patient was admitted after being struck by a vehicle. The resultant injuries required that her femur be externally fixed as seen in the images below to allow fo


This Week in Plastics - Keloids, VAC

In addition to the OR, I have been able to see many very interesting cases during office hours. The nurse that works with Dr. Spector in office hours has shown me where many of the instruments and supplies are stored allowing me to assist Dr. Spector and be more involved with the care of the patient. One patient that has been coming in twice a week for V.A.C. changes has a very persistent wound.  The patient had a hernia repair which left a large opening in his abdominal area. A hernia occurs when underlying tissue protrudes through tissue that usually encloses it (generally muscle). As in the diagram the intestines may break through the abdominal muscle wall and require repair (picture from: http://www.pbennett.com.au/images/abdominal_hernia.jpg). In the picture on the right, you can see the black sponge that has been placed over the open wound of a patient that is used with the V.A.C. system to aid in closure (picture from: http://www.emedicine.com/med/images/4884med3722-30 .jpg). The V.A.C. has helped to close the wound, but at a very slow rate and each week during office hours Dr. Spector debrides the wound of dead tissues. It is interesting to see the progress of the patient’s wound closure with the help of a simple suction machine. Many of the cases during office hours are post-operative check-ups, but occasionally there are pre-operative consults.
The patient had a hernia repair which left a large opening in his abdominal area. A hernia occurs when underlying tissue protrudes through tissue that usually encloses it (generally muscle). As in the diagram the intestines may break through the abdominal muscle wall and require repair (picture from: http://www.pbennett.com.au/images/abdominal_hernia.jpg). In the picture on the right, you can see the black sponge that has been placed over the open wound of a patient that is used with the V.A.C. system to aid in closure (picture from: http://www.emedicine.com/med/images/4884med3722-30 .jpg). The V.A.C. has helped to close the wound, but at a very slow rate and each week during office hours Dr. Spector debrides the wound of dead tissues. It is interesting to see the progress of the patient’s wound closure with the help of a simple suction machine. Many of the cases during office hours are post-operative check-ups, but occasionally there are pre-operative consults.
In addition to consults, Dr. Spector also performs minor procedures during office hours. A patient last week that had a severe case a keloids was treated with steroid injections. Keloids are collections of scar tissue that form after an injury or surgery. Keloids are cosmetically unappealing and are often quiet painful. In the picture below, you can see an example of a keloid (picture from: http://fig.cox.miami.edu/~cmallery/150/protein/keloid.jpg).  In this case the keloids had formed after a spinal surgery which meant they were in a very uncomfortable position. Two treatments for keloids that show promise are compression and steroid injections. It is not completely clear how the steroids aid in smoothing out the keloids, but with the injections the patient can see definite progress in both the appearance and pain level of the keloids.
In this case the keloids had formed after a spinal surgery which meant they were in a very uncomfortable position. Two treatments for keloids that show promise are compression and steroid injections. It is not completely clear how the steroids aid in smoothing out the keloids, but with the injections the patient can see definite progress in both the appearance and pain level of the keloids.
Learning to Tie Sutures
The other day it was relatively slow all around and so I was fortunate enough to sit down with some doctors and doctors-to-be and learned the subtle art of suturing. I’m sure we have all witnessed trained surgeons throwing knots like there’s no tomorrow; however, rather surprisingly there is a lot that goes into even a simply stitch.
So apparently, there are over 1400 different types of knots (or at least according to the Encyclopedia of Knots…who knew there was even such an encyclopedia…shows I was never Eagle Scout extraordinaire). Anyway, despite the plethora of knots, surgeons only use several different ones. The most common are as follows: square knot, surgeon’s knot, deep tie,
instrument tie, running stitch and ligation with a hemostat (a surgical clamp).
 The square knot is essentially the same thing as the knot you use to tie your shoelaces, with the exception that, like everything in medicine, you have to make it more complicated than necessary. Similarly, the surgeon’s knot is essentially a double knot. The deep tie is usually used to approximate tissues deep in a cavity. The instrument tie is a quick way to tie a knot by simply wrapping the suture around the needledriver and then using needledriver to pull the other end of the suture through the loop you just made. Both square and surgeon’s knots can be thrown in this manner. The running stitch is just a continuous series of stitches thrown with one suture. I didn’t learn how to do a ligation tie, but I’ve seen it done a few times. It seems like you clamp down a vessel, loop the suture around the hemostat and tie off the vessel behind the clamp with some square knots.
The square knot is essentially the same thing as the knot you use to tie your shoelaces, with the exception that, like everything in medicine, you have to make it more complicated than necessary. Similarly, the surgeon’s knot is essentially a double knot. The deep tie is usually used to approximate tissues deep in a cavity. The instrument tie is a quick way to tie a knot by simply wrapping the suture around the needledriver and then using needledriver to pull the other end of the suture through the loop you just made. Both square and surgeon’s knots can be thrown in this manner. The running stitch is just a continuous series of stitches thrown with one suture. I didn’t learn how to do a ligation tie, but I’ve seen it done a few times. It seems like you clamp down a vessel, loop the suture around the hemostat and tie off the vessel behind the clamp with some square knots.
Anyway, this post was not meant to reveal anything profound, I just thought it was an interesting experience to share and some potentially useful knowledge to know…next time I need to tie off my blood vessels I will know…and knowing is half the battle…
Phase Contrast MRI 2: The Revenge
Last week, I talked briefly about how we use successive slices in a cine MRI sequence to evaluate heart function. Using this type of image analysis, we are able to find information such as cardiac output and ejection fraction, as well as volume and mass information during various stages of the heart. In addition, we can use a technique called Phase Contrast MRI to obtain flow information. While the data obtained from Phase Contrast MRI is slightly different than the data obtained from segmenting a cine MRI, it is possible to correlate the two and from this comparison, we can determine the agreement between our two measurements.
Phase contrast works by exploiting the nature of MR imaging. Two gradients, with equal magnitude but opposite orientation, are applied to the area of interest in rapid succession. Objects that remain completely stationary undergo no net phase shift, as the opposing gradients “cancel out” the effects of one another. However, objects that are moving undergo a change in phase proportional to their velocity. This concept is illustrated in the diagram below.

Phase Contrast gradient fields on stationary and moving objects
(image obtained from radiographics.rsnajnls.org)
The top panels demonstrate the gradients on a stationary object. The first gradient “tips” the atoms by varying amounts, depending on their location within the gradient field. These tip angles are eliminated upon the application of the second gradient field. The end result gives the effect as if no gradient was applied at all, and all magnetic moments are uniform and in a single direction.
On the other hand, a moving object will undergo a predictable change in phase. The bottom panels illustrate what happens when a single atom travels linearly through the same region. The end result is a phase distinctly different than that of the stationary atoms.
This information can be visualized by viewing the phase information of a particular MR image sequence. Typically, a neutral gray color is assigned to pixels in which there is no net phase difference. Black indicates that the objects within the pixel are moving towards the viewer, while a white color indicates that objects are moving away. Varying velocities are indicated by the intensity of these shapes. From this, we can quickly determine the direction and amount of flow. Further, by using computer automation and manual segmentation, we can determine the amount of flow in specific areas.
In this study, we will be performing phase contrast analyses of the aorta. By quantifying the amount of blood flowing through the aorta, we can indirectly determine the stroke volume of the heart, which will provide an additional form of “ground truth” by which we can further evaluate our automatic segmentation algorithm.

Magnitude MR image ("Regular")
(image obtained from radiographics.rsnajnls.org)

Phase Contrast MR image (new and improved!)
(image obtained from radiographics.rsnajnls.org)